
September 15, 2020
PHC Corporation
2-38-5 Nishishimbashi, Minato-ku, Tokyo 105-8433, Japan
JCR Pharmaceuticals Co., Ltd.
3-19 Kasuga-cho, Ashiya, Hyogo 659-0021, Japan
Tokyo and Hyogo, September 15, 2020 - PHC Corporation (headquarters: Minato-ku, Tokyo, President: Kyoji Morimoto, hereafter PHC) and JCR Pharmaceuticals Co., Ltd. (headquarters: Ashiya, Hyogo, Chairman and President: Shin Ashida, hereafter JCR) announce a dedicated smartphone application for the motorized growth hormone delivery device GROWJECTOR® L*1 that the two companies have jointly developed. This application will be provided to the Tokyo Medical and Dental University (Bunkyo-ku, Tokyo) and other medical institutions for clinical research starting September 2020.
The application will be provided for clinical research in the following area:
“Exploratory study on the effect on medication adherence*2 for short-stature children when using new growth hormone liquid formulation aimed to reduce injection irritation” (Project Leader: Dr. Kenichi Kashimada of Tokyo Medical and Dental University Graduate School of Medical and Dental Sciences, Department of Pediatrics and Developmental Biology).
GROWJECTOR® L is a motorized drug delivery device manufactured and sold by PHC, that is used exclusively for the recombinant human growth hormone GROWJECT® subcutaneous 6mg and 12mg*3 treatment that is manufactured and sold by JCR. The device records patient injections (dosing history), which are stored in the device memory. Using the dedicated application, the dosing history stored in GROWJECTOR® L can be wirelessly transmitted to a smartphone, allowing patients to more easily check the details of a specific injection, as well as review detailed dosing history including the time and number of injections.
Previously, gathering accurate data had been difficult, as it was dependent on patient memory, as well as recorded information. The planned clinical research will use the application to accurately collect basic data related to the detailed dosing history (adherence rate), and is expected to provide useful knowledge on the correlation between irritation during injection and maintaining on treatment, as well as improving medication adherence in growth hormone therapy.
The application has already been provided to the clinical research program “Effectiveness of smartphone-based mobile application on medication adherence in patients receiving growth hormone therapy - An exploratory observation study (UMIN000035997)”*4 (Project Leader: Dr. Tatsuhiko Urakami of Nihon University Hospital) that is aiming to gain information for practical use. Data collection for this research program has completed, and the application is planned for Google Play release in Japan this October.
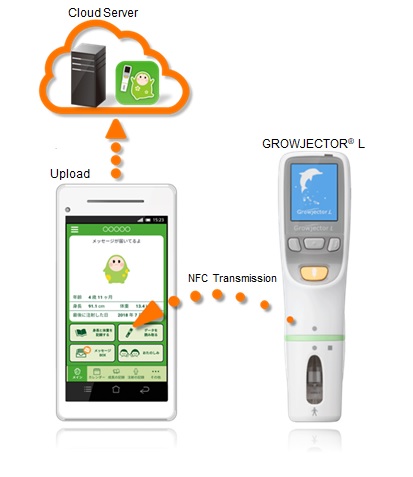 Application and GROWJECTOR® L linkage
Application and GROWJECTOR® L linkage
*1 Official generic term: Drug/vaccine injector
*2 Accepting the disease and positively receiving treatment using drugs with doctor’s instructions.
*3 Approved indications of GROWJECT® 6mg/GROWJECT®12 mg
- Growth Hormone Deficiency
- Turner Syndrome
- Adult Growth Hormone Deficiency
- Small for Gestational Age
Please visit JCR website for further information: jcrgh.com/ (Japanese website)
*4 Clinical research that began in March 2019 and currently ongoing at Department of Pediatrics, Nihon University Hospital. Aims to study patient dosing with growth hormone using dedicated smartphone application for GROWJECTOR® L, and analyze factors etc. related to reduced adherence in growth hormone therapy.
rctportal.niph.go.jp/en/detail?trial_id=UMIN000035997
About PHC Corporation
Established in 1969, PHC Corporation is a Japanese subsidiary of the PHC Group, a global healthcare company group that develops, manufactures, sells, and services healthcare solutions across diabetes management, diagnostics and life sciences, as well as providing healthcare service businesses. Committed to its corporate mission, “We contribute to the health of society through our diligent efforts to create healthcare solutions that have a positive impact and improve the lives of people,” the company offers value-added products and services to customers in over 125 countries. The IVD Business Unit has worked to improve medical services for patients by contributing to early diagnosis and efficient medical care for diseases such as diabetes and asthma through its development, manufacture, and distribution of testing and analyzing devices including blood glucose monitoring systems.
www.phchd.com/global
About JCR Pharmaceuticals Co., Ltd
JCR is a specialty pharma company engaged in research, development, manufacturing, and marketing of biopharmaceuticals and regenerative medicine with a focus on rare diseases. Its philosophy, “Contributing towards people’s healthcare through pharmaceutical products” drives JCR to create innovative pharmaceutical products as value-added treatment options for the under-served patient populations.
www.jcrpharm.co.jp/en/site/en/index.html
Media contact
Corporate Communications Group
PHC Holdings Corporation
+81-3-6778-5311
Investors and Media
JCR Pharmaceuticals Co., Ltd.
Email: ir-inrfo@jcrpharma.co.jp