Freezers and Refrigerators for CDC Compliant Vaccine Storage
Benchtop, undercounter, upright single door, double door and combo models


Vaccine potency relies on protection against temperature extremes through proper storage at every level of the cold chain. While the focus of temperature control is often on overheating, vaccines can be just as susceptible to freezing risks. Learn how to protect your vaccines with this free ebook.
|
|
PHCbi brand refrigerators and freezers are engineered from the inside out to create, maintain and restore precise temperatures that are not commonly found with household or commercial cabinets.
|
Medical Grade Pharmacy, Biomedical and Laboratory Refrigerators and Freezers
- Refrigerators incorporate specifically designed refrigeration systems, protecting vaccines from freezing
- Forced air systems provide close top-to-bottom uniformity at all shelf and drawer levels
- Microprocessor controls assure precise temperature throughout the chamber
- Easy to view control panel displays with alarm systems that have access to remote monitoring
- Reserve cooling for rapid temperature recovery following door openings
- Tolerance for high ambient temperature with high performance refrigeration systems and CFC free insulation
Vaccine Storage Recommendations
If a vaccine freezes, its efficacy may be diminished or destroyed without visible indication to administering clinicians. Thus, vaccines inadvertently frozen in cold chain distribution or local storage may not offer protection to patients.1
In an effort to assure the quality of vaccines at the end of the distribution cold chain, the United States Centers for Disease Control and Prevention (CDC) has published guidelines for best practices in vaccine storage.2 These guidelines, which continue to evolve, are based on studies conducted by the National Institute of Standards and Technology (NIST).3
These guidelines, established in 2014, were designed to isolate and identify the conditions that affect vaccine efficacy and storage integrity, including the following areas of focus:
- Must offer dedicated storage in a stand-alone refrigerator specifically designed to establish and maintain key performance parameters.
- Dormitory-style units should not be used under any circumstances. This type of refrigerator poses a significant risk of freezing a vaccine even when it is used for temporary storage. A NIST report showed that this type of unit demonstrated inconsistent temperature control, regardless of where the vaccine was located in the chamber. Within 2 weeks of use, median temperature of the refrigerator setpoint had drifted approximately 4°C lower, freezing the vaccines contained inside.4
- Must maintain accurate, uniform and repeatable storage temperature over a range of 2°C to 8°C. The PHC Corporation factory setpoint is 5°C.
- Must protect from freezing temperatures anywhere in the refrigerator. This is critical for storage of small vaccine doses that can freeze quickly.
- Must be monitored by a NIST calibrated thermometer, documented with a certificate of calibration. The CDC recommended uncertainty is +/- 0.5°C.
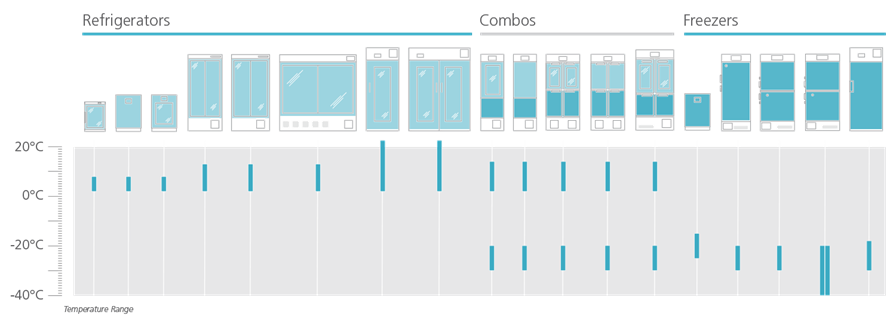
PHCbi refrigerators, freezers and combo refrigerators/freezers are available in a range of cabinet sizes and operating temperatures. This graphic index illustrates the relationship among the high performance product family.
Refrigerators

|
High Performance Undercounter Refrigerator
3.0 cu.ft. (85 L)
|

|
High Performance Undercounter Refrigerator
5.0 cu.ft. (142 L)
|

|
High Performance Undercounter Refrigerator
5.0 cu.ft. (142 L)
|

|
High Performance ECO Pharmaceutical Refrigerator
12.2 cu.ft. (345 L)
|

|
High Performance Medical Refrigerator
19.5 cu.ft. (554 L)
19.4 cu.ft. (550 L)
|

|
Pharmaceutical Refrigerator
24.2 cu.ft. (684 L)
23.7 cu.ft. (671 L)
|

|
Pharmaceutical Refrigerator
40.8 cu.ft. (1155 L)
|

|
Pharmaceutical Refrigerator
41.1 cu.ft. (1165 L)
|

|
Pharmaceutical Refrigerator
48.2 cu.ft. (1033 L)
48.0 cu.ft. (1029 L)
|
Refrigerators/Freezers Combos

|
Pharmaceutical Refrigerator with Freezer
R: 6.3 cu.ft. (179 L)
F: 2.8 cu.ft. (80 L)
|

|
Pharmaceutical Refrigerator with Freezer
R: 6.3 cu.ft. (179 L)
F: 2.8 cu.ft. (80 L)
|

|
Pharmaceutical Refrigerator with Freezer
F: 11.5 cu.ft. (326 L)
R: 4.8 cu.ft. (136 L)
|

|
Pharmaceutical Refrigerator with Freezer
F: 11.5 cu.ft. (326 L)
R: 4.8 cu.ft. (136 L)
|

|
Pharmaceutical Refrigerator with Freezer
F: 14.7 cu.ft. (415 L)
R: 6.2 cu.ft. (176 L)
|
Freezers

|
Undercounter Medical Freezer
5.0 cu.ft. (142 L)
|

|
Biomedical ECO Freezer
13.0 cu.ft. (369 L)
|

|
Biomedical ECO Freezer
16.9 cu.ft. (479 L)
|

|
Biomedical ECO Freezer
17.8 cu.ft. (504 L)
|

|
Biomedical Freezer
22.0 cu.ft. (623 L)
24.4 cu.ft. (690 L)
|
About PHC Corporation of North America
PHC Corporation of North America, located in Wood Dale, IL, is a leader in laboratory equipment for the biopharmaceutical, life sciences, academic, healthcare and government markets. Product lines under the PHCbi brand include the space saving and energy efficient VIP® ECO and TwinGuard® ultra-low temperature freezers, cryogenic and biomedical freezers, pharmacy and high-performance refrigerators, cell culture CO2 and multigas incubators, and Drosophila/plant growth chambers. PHC Corporation of North America is a subsidiary of PHC Holdings Corporation, Tokyo, Japan, which is a global healthcare company that develops, manufactures, sells, and services solutions across diabetes management, healthcare solutions, diagnostics and life sciences.
1 A 2012 Centers for Disease Control and Prevention (CDC) report revealed improper storage of some vaccines from the Vaccines for Children (VFC) program serving an estimated 40 million children through a national network.
2 NSF International (NSF) and The U.S. Center for Disease Control and Prevention have assigned industry consultants comprised of leading refrigeration and freezer cabinet manufacturers, including PHC Corporation of North America, to establish vaccine storage guidelines with an emphasis on establishing criteria for storage refrigerators to protect refrigerated vaccines from freezing. These criteria will require that all vaccines be stored in refrigerators that meet performance standards currently offered by PHC.
4 U.S. National Institute of Standards and Technology. (2009).
Thermal analysis of refrigeration systems used for vaccine storage. Gaithersburg, MD: Chojnacky, M., Miller, W., Ripple, D., & Strouse, G. Retrieved from
http://www.nist.gov/customcf/get_pdf.cfm?pub_id=904574